Popular on s4story
- USA Best Book Awards Finalist What Love Leaves Behind Releases March 24 - 422
- Oberfeld Press Author Releases Typographic Series Ahead of July Publication - 204
- The World's First Fully Regenerative Economy: Securing Energy, Food, and a Clean Planet - 161
- New Book Synthesizes Six Peer-Reviewed Research Programs Into Unified Framework for Consciousness - 103
- QuickTrack by Datalex Transforms Retail Promoter Management with Claude AI and Real-Time Insights
- High5VR Announces World's First Fully Immersive First-Person VR Movie
- New YA Fantasy "The Whispering Key" by Auren Keyes Launches The Luminara Chronicles
- Distributed Social Media - Own Your Content
- Progressive Dental & The Closing Institute Partner with Zest Dental Solutions to Elevate Full-Arch Growth and Patient Outcomes
- P-Wave Classics to publish Robert Bage's Hermsprong in three volumes, beginning 12 May
Similar on s4story
- FinIQ Edu Launches High-Impact Workshop Vertical to Close the Workplace Benefits Gap—Drives 82% Surge in 401(k) Participation Intent
- HousingWire launches Mortgage Rankings, bringing a data-driven benchmark to originator performance
- Registered Nurse Launches Healthcare Wealth Strategy Practice for Healthcare Professionals
- Just 1 in 57 Crypto Owners Globally Pay Taxes on Their Holdings, New Report Finds
- IQSTEL accelerates toward profitability inflection with $317M revenue and AI-driven expansion; IQSTEL Inc. (N A S D A Q: IQST) i
- AI-Driven Breakthrough Unleashed: Bionic Intelligence Platform Goes Live to Capture Massive Biotech Opportunity: KALA BIO, Inc. (N A S D A Q: KALA)
- Surging Into Hyper-Growth Mode With Record Revenue, Raised 2026 Guidance, and Game-Changing AI Platform; Off The Hook YS (NYSE American: OTH)
- Daniel Kaufman Expands Kaufman & Company Real Estate Platform With New Acquisitions, AI-Driven Industrial Development and Nationwide Growth Initiative
- purelyIV Launches Lab Testing Services in Metro Detroit
- NRx Pharmaceuticals Could Be on the Verge of a Breakout Year as AI, FDA Catalysts, and Mental Health Demand Converge
$300 Million in Milestones Plus Tiered Double-Digit Royalties to License & Distribute NRX-100 Drug: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
S For Story/10653530
$NRXP is Poised to Address the $3 Billion + Suicidal Depression Market in the US
MIAMI - s4story -- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
New Drug Application for Treatment of Suicidal Depression; Planned NDA for Accelerated Approval for Bipolar Depression in People at Risk of Akathisia.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market
NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year. This represents a $3-5 billion market at expected pricing. Based on the data in the trials referenced above, the Company's regulatory counsel encouraged the Company to file an NDA for suicidal depression for NRX-100.
In a January 2025 report, respected investment analysis firm D. Boral Capital assigned NRXP a $31 Price Target. The full report may be viewed at this link: https://www.nrxpharma.com/wp-content/uploads/2025/01/HOPE-Therapeutics-Advances-With-Kadima.pdf.
More on S For Story
Fourth Quarter and Full Year 2024 Financial Results and Provides Corporate Update
On March 17th NRXP announced its financial results for the quarter and year ended December 31, 2024, and provided a business update. The announcement included the following key highlights:
NRXP initiated filing of a New Drug Application ("NDA") to the FDA for NRX-100 (IV Ketamine) for the treatment of Suicidal Depression; planned filing of an NDA for Accelerated Approval under Breakthrough Designation and Priority Review of NRX-101 for the treatment of bipolar depression in people at risk of akathisia. Both have anticipated PDUFA dates prior to December 31, 2025
NRXP has accepted non-binding potential terms from a commercial pharmaceutical company to license and distribute NRX-100, providing over $300 million in milestones plus tiered double-digit royalties based on net sales
NRXP retained a leading regulatory law firm to file a citizen's petition with the US Food and Drug Administration ("FDA") to remove benzethonium chloride – a toxic preservative -- from presentations of ketamine intended for intravenous use; planned 2Q25 filing of an Abbreviated New Drug Application ("ANDA") for the use of preservative-free ketamine in all current indications
Wholly owned subsidiary HOPE Therapeutics, signed non-binding letters of intent to acquire three precision psychiatry centers and is currently completing financial due diligence and definitive agreements. Currently negotiating the terms for the acquisition of six additional centers
The HOPE acquisitions are planned to form the foundation for a national network offering interventional psychiatry services to treat suicidal depression, post-traumatic stress disorder ("PTSD") and related conditions
NRXP received and negotiating a term sheet from a publicly-traded strategic investor currently engaged in manufacturing Transcranial Magnetic Stimulation ("TMS") devices to provide capital in support of expansion of further HOPE clinic acquisitions.
NRXP has engaged BTIG as financial advisor for clinic acquisition and capital formation; leading global financial services firm specializing in investment banking, institutional trading, research, and related brokerage services for strategic growth opportunities.
NRXP regained compliance with the NASDAQ market value of listed securities ("MVLS") requirement.
Substantially reduced operating costs compared to prior year
Management continues to forecast, although no assurances can be given, profitability on a forward-looking run-rate basis by year end 2025
More on S For Story
NRXP filed Module 3 (manufacturing) of its New Drug Application ("NDA") for NRX-100 (preservative-free sterile IV ketamine) in a tamper-resistant, diversion resistant packaging presentation in the fourth quarter of 2024. NRX-100 was previously granted Fast Track Designation by FDA in combination with use of NRX-101. Ketamine efficacy data from four clinical trials are intended to support the filing. Three manufacturing lots are now complete, with filed stability data suitable for shelf life exceeding two years at room temperature. The anticipated PDUFA date for this NDA is prior to December 31, 2025.
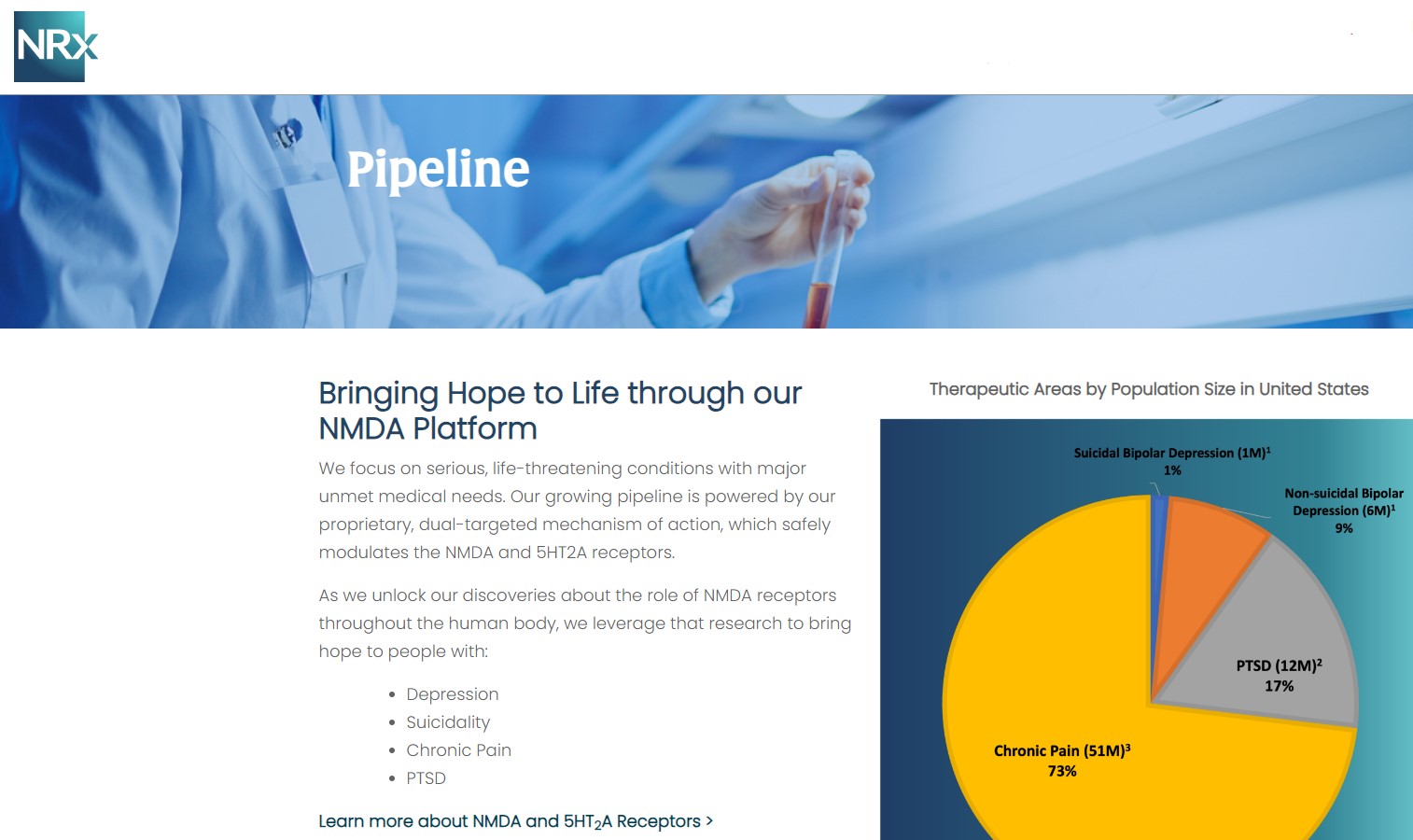
NRX-100 is poised to address the over $3 billion Suicidal Depression market in the US.
NRXP has accepted non-binding potential terms from a commercial pharmaceutical company to license and distribute NRX-100 (preservative-free IV ketamine) that provides for over $300 million in potential milestones plus a tiered double-digit royalty, subject to further due diligence and finalized agreement.
NRXP announced its intention to file an NDA for Accelerated Approval under Breakthrough Designation and Priority Review of NRX-101 for the treatment of bipolar depression in people at risk of akathisia, based on the Phase 2b/3 and STABIL-B data. Three manufacturing lots are now complete with more than 12 months of room temperature shelf-stability. The anticipated PDUFA date for this application is prior to December 31, 2025.
NRXP estimates that the market for the initial indication is over $2 billion, while the broad bipolar market could exceed $5 billion.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
New Drug Application for Treatment of Suicidal Depression; Planned NDA for Accelerated Approval for Bipolar Depression in People at Risk of Akathisia.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market
NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year. This represents a $3-5 billion market at expected pricing. Based on the data in the trials referenced above, the Company's regulatory counsel encouraged the Company to file an NDA for suicidal depression for NRX-100.
In a January 2025 report, respected investment analysis firm D. Boral Capital assigned NRXP a $31 Price Target. The full report may be viewed at this link: https://www.nrxpharma.com/wp-content/uploads/2025/01/HOPE-Therapeutics-Advances-With-Kadima.pdf.
More on S For Story
- Just 1 in 57 Crypto Owners Globally Pay Taxes on Their Holdings, New Report Finds
- IQSTEL accelerates toward profitability inflection with $317M revenue and AI-driven expansion; IQSTEL Inc. (N A S D A Q: IQST) i
- AI-Driven Breakthrough Unleashed: Bionic Intelligence Platform Goes Live to Capture Massive Biotech Opportunity: KALA BIO, Inc. (N A S D A Q: KALA)
- Surging Into Hyper-Growth Mode With Record Revenue, Raised 2026 Guidance, and Game-Changing AI Platform; Off The Hook YS (NYSE American: OTH)
- Mom Creators Coalition Launches with WaterWipes® as Official Founding Sponsor
Fourth Quarter and Full Year 2024 Financial Results and Provides Corporate Update
On March 17th NRXP announced its financial results for the quarter and year ended December 31, 2024, and provided a business update. The announcement included the following key highlights:
NRXP initiated filing of a New Drug Application ("NDA") to the FDA for NRX-100 (IV Ketamine) for the treatment of Suicidal Depression; planned filing of an NDA for Accelerated Approval under Breakthrough Designation and Priority Review of NRX-101 for the treatment of bipolar depression in people at risk of akathisia. Both have anticipated PDUFA dates prior to December 31, 2025
NRXP has accepted non-binding potential terms from a commercial pharmaceutical company to license and distribute NRX-100, providing over $300 million in milestones plus tiered double-digit royalties based on net sales
NRXP retained a leading regulatory law firm to file a citizen's petition with the US Food and Drug Administration ("FDA") to remove benzethonium chloride – a toxic preservative -- from presentations of ketamine intended for intravenous use; planned 2Q25 filing of an Abbreviated New Drug Application ("ANDA") for the use of preservative-free ketamine in all current indications
Wholly owned subsidiary HOPE Therapeutics, signed non-binding letters of intent to acquire three precision psychiatry centers and is currently completing financial due diligence and definitive agreements. Currently negotiating the terms for the acquisition of six additional centers
The HOPE acquisitions are planned to form the foundation for a national network offering interventional psychiatry services to treat suicidal depression, post-traumatic stress disorder ("PTSD") and related conditions
NRXP received and negotiating a term sheet from a publicly-traded strategic investor currently engaged in manufacturing Transcranial Magnetic Stimulation ("TMS") devices to provide capital in support of expansion of further HOPE clinic acquisitions.
NRXP has engaged BTIG as financial advisor for clinic acquisition and capital formation; leading global financial services firm specializing in investment banking, institutional trading, research, and related brokerage services for strategic growth opportunities.
NRXP regained compliance with the NASDAQ market value of listed securities ("MVLS") requirement.
Substantially reduced operating costs compared to prior year
Management continues to forecast, although no assurances can be given, profitability on a forward-looking run-rate basis by year end 2025
More on S For Story
- BookCAMP 2026 Announces Schedule: A Premier Gathering for Independent Authors and Publishers
- PandaGuarantee Launches Rent Guarantor Service in New York City
- The $112M Marketing Lesson Joe Whyte Learned: Why 'More Traffic' Is the Biggest Lie in Digital Marketing
- Daniel Kaufman Expands Kaufman & Company Real Estate Platform With New Acquisitions, AI-Driven Industrial Development and Nationwide Growth Initiative
- purelyIV Launches Lab Testing Services in Metro Detroit
NRXP filed Module 3 (manufacturing) of its New Drug Application ("NDA") for NRX-100 (preservative-free sterile IV ketamine) in a tamper-resistant, diversion resistant packaging presentation in the fourth quarter of 2024. NRX-100 was previously granted Fast Track Designation by FDA in combination with use of NRX-101. Ketamine efficacy data from four clinical trials are intended to support the filing. Three manufacturing lots are now complete, with filed stability data suitable for shelf life exceeding two years at room temperature. The anticipated PDUFA date for this NDA is prior to December 31, 2025.
NRX-100 is poised to address the over $3 billion Suicidal Depression market in the US.
NRXP has accepted non-binding potential terms from a commercial pharmaceutical company to license and distribute NRX-100 (preservative-free IV ketamine) that provides for over $300 million in potential milestones plus a tiered double-digit royalty, subject to further due diligence and finalized agreement.
NRXP announced its intention to file an NDA for Accelerated Approval under Breakthrough Designation and Priority Review of NRX-101 for the treatment of bipolar depression in people at risk of akathisia, based on the Phase 2b/3 and STABIL-B data. Three manufacturing lots are now complete with more than 12 months of room temperature shelf-stability. The anticipated PDUFA date for this application is prior to December 31, 2025.
NRXP estimates that the market for the initial indication is over $2 billion, while the broad bipolar market could exceed $5 billion.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
0 Comments
Latest on S For Story
- SelfCare is now HealthCare across America
- "King of Cool" now on audio & Kindle—A Steve McQueen Devotional: Bullitt Points to God Vol. I & II
- Homeowner Prep Announces Strategic Language Shift: Replacing "Renters" with "Future Homeowners" to Inspire Wealth-Building Mindsets
- LiposoMore™ Redefines Bioavailability: Joyful Nutritional Launches High-Performance Liposomal Vitamin C and Iron for the Global Supplement Market
- GDE Tree Services Expands Operations into Sydney, NSW
- Tuckwell Machinery Expands CNC Range to Support Australian Cabinet Makers
- Atlanta Teen Author and #1 Top New Release Sarai Lokey Takes "Holy Rebel" Straight To The Top!
- The Inner Power of Emotional Self-Leadership
- Dr. Nadene Rose Shares the Secret to True Success: Faith, Obedience, and Divine Purpose
- eBook Publishing Services in USA California Book Publishers Helps Authors Turn Ideas into Successful
- Understanding Unexpected Death: Why Independent Autopsies Matter in Cases Without Clear Cause
- Epic Pictures Group Sets North American Release Date for the Thriller NO ORDINARY HEIST
- eBook Ghostwriting Services in USA: Noble Book Publishers Helps Authors Turn Ideas into Bestsellers
- Award-Winning REALTOR® Paige Coker Joins Corcoran DeRonja Real Estate
- Over 98% of crypto owners globally don't declare taxes, new report find
- TicTac Group acquires French EdTech company Distrisoft
- Equestrian Expert Rominger Receives National Recognition Through the INDEPENDENT PRESS AWARD®!
- A Story of Loyalty and Loss: Boone Forever Reflects on the Dog That Changed One Man's Life
- Mark Dobosz Makes Donorassess.org Free To Every Nonprofit On The Planet
- Genpak Announces Closure of Utah Manufacturing Facility