Popular on s4story
- Open Art Call | The Art of Artificial Intelligence | Copenhagen - 281
- Dylan Johnson Releases New Book - The Manipulator's Gambit - 251
- Premieres of 10th Annual NY Dog Film Festival & 8th Annual NY Cat Film Festival on Sunday, October 26, 2025 to Benefit Animal Lighthouse Rescue - 248
- Entry Level Acting in LA 2025 Workbook to be Released in West Hollywood, California USA 2pm 10/11/25 - 244
- Leading Digital Finance Platform YNQTL Launches Revolutionary Web3 Digital Asset Trading Platform - 241
- 3E Launches First AI Agent Designed to Respond with Empathy for College Recruitment - 241
- IDCXS Addresses Crypto Trading Pain Points with 2 Million TPS Processing and Multi-Layer Security Architecture - 237
- 120% Revenue Surge with Four Straight Profitable Quarters Signal a Breakout in the Multi-Billion Dollar Homebuilding Market: Innovative Designs $IVDN - 235
- Mamta Jha Mishra Releases New Book: "Energy Management: Mantras for Caregivers" - 230
- New Book Reveals 10 Simple "Stress Hacks" Hidden in the Human Body - 224
Similar on s4story
- Essential Living Support Opens First VA Medical Foster Home in Cheyenne, Wyoming
- $150 Million Financing Initiates N A S D A Q's First Tether Gold Treasury Combining the Stability of Physical Gold with Blockchain $AURE
- What If Help Could Come Before the Fall?
- Coming Up this Weekend on CNBC Mike Milligan Joins Tom Hegna on "Financial Freedom with Tom Hegna"
- $73.6M Pipeline, $10M Crypto Play & Legal Firepower: Why Investors Are Watching Cycurion (N A S D A Q: CYCU) Like a Hawk
- QView Medical and Eve Wellness Announce Partnership to Advance Direct-to-Consumer Breast Cancer Screening With FDA-Approved AI
- NASA Agreements, New Ocean Exploration Applications Added to Partnerships with Defiant Space Corp and Emtel Energy USA for Solar Tech Leader: $ASTI
- Chic and Secure: The Blue Luna Debuts Stylish Keychains with Purpose
- $500,000 in Stock Dividend for Shareholders in 2025 Sweetens The Pot on Success of Becoming Debt Free with No Convertible Notes or Warrants for $IQST
- Cancer Survivor Roslyn Franken Marks 30-Year Milestone with Empowering Gift for Women Survivors
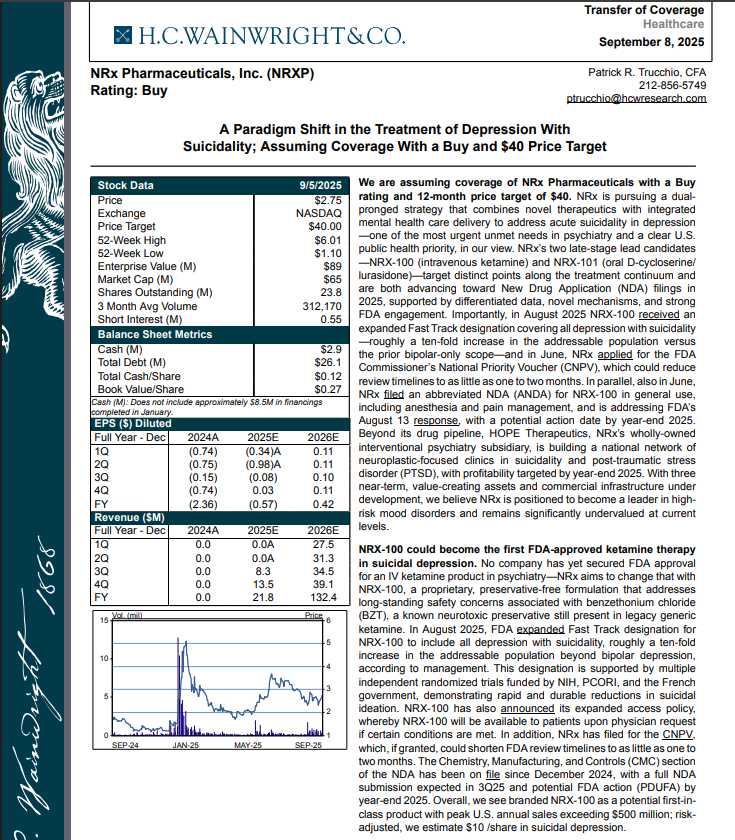
$40 Price Target for $NRXP in H. C. Wainright Analyst Report on Leader in $3 Billion Suicidal Depression Market with Superior NRX 100 Drug Therapy
S For Story/10671274
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP Continues Expansion with Completion of Dura Medical Acquisition in Network of Interventional Psychiatry Clinics
MIAMI - s4story -- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
H.C. Wainright Analyst Report Cites Paradigm Shift in the Treatment of Depression With Suicidality; Assuming Coverage with Buy and $40 Price Target.
Dura Medical Acquisition Completed in Network of Interventional Psychiatry Clinics.
FDA Fast Track Designation for NRX 100 for Suicidal Ideation in Patients with Depression, Including Bipolar Depression.
Designation Includes an FDA Determination That NRX-100 has Potential to Address an Unmet Need.
Actions Taken to Request the Removal of Benzethonium Chloride from Ketamine Products in Favor of the Company's Safer and Superior Options.
$7.8 Million Debt Financing to Fuel NRXP HOPE Clinic Acquisitions with
Universal Capital, LLC.
Accepted Non-Binding Potential Terms to License and Distribute NRX-100 Drug Providing Over $300 Million in Milestones Plus Tiered Double-Digit Royalties.
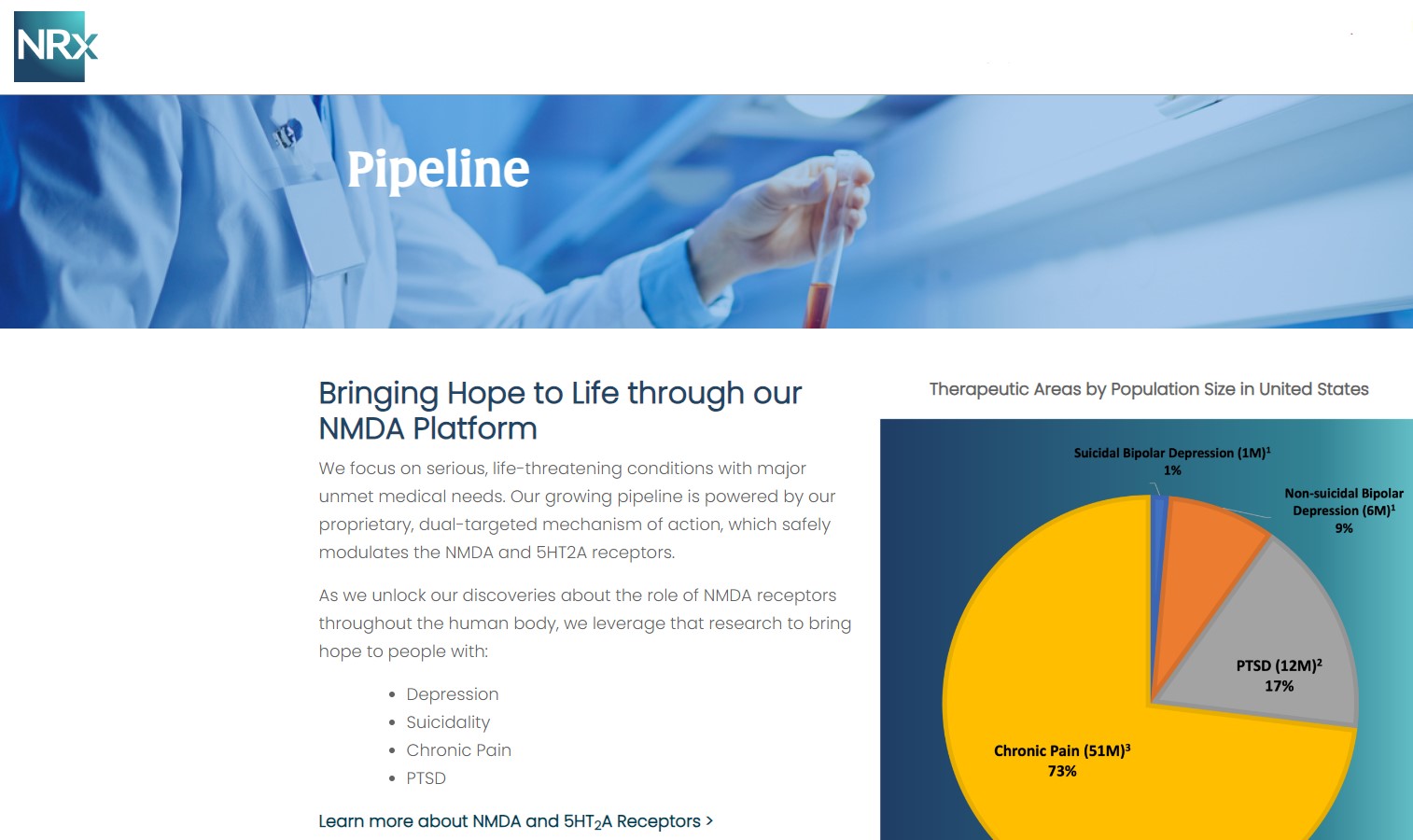
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
On September 8th H.C. Wainwright has issued a new Analyst Report on NRXP: "A Paradigm Shift in the Treatment of Depression With Suicidality" Assuming Coverage With a Buy and $40 Price Target. The full report may be accessed at this direct link: https://hcwco.bluematrix.com/links2/secure/pdf/acdd3260-630e-48e6-9c2f-03fbc0be37d6
More on S For Story
Dura Medical Acquisition Completed in Network of Interventional Psychiatry Clinics
On September 8th NRXP announced the closing of its acquisition of Dura Medical. Dura, together with the pending Neurospa TMS and Cohen and Associates acquisitions, are planned to provide a comprehensive service offering to patients at more than 8 locations along the West Coast of Florida. Dura is revenue generating and EBITDA positive.
Dura delivers a full range of precision psychiatry services for severe depression and PTSD, including Ketamine Therapy and Transcranial Magnetic Stimulation to Veterans and civilian patients.
Expanded Access Policy for NRX-100 (preservative-free ketamine)
On August 27th NRXP announced its expanded access policy for NRX-100 (preservative-free ketamine) based on grant of Fast Track designation for NRX-100 in the treatment of suicidal ideation in patients with depression, including bipolar depression.
In granting the Fast Track designation, FDA made the determination that NRXP NRX-100 has the potential to address an unmet need, based on an assessment of the preliminary data contained in the Fast Track designation request. Accordingly, NRXP NRX-100 is available for expanded access to eligible patients.
Second Quarter 2025 Update
On August 18th NRXP announced financial results for the quarter ended June 30, 2025, and provided a corporate update. As of June 30, 2025, NRXP had approximately $2.9 million in cash and cash equivalents.
Drug Development
Grant of expanded Fast Track Designation for NRXP NRX-100 from the FDA for all indications and types of depression and related disorders based on its potential to satisfy an unmet medical need.
Approximately 10-fold expansion of the addressable market to 13 million Americans, compared to the original Fast Track Designation issued in 2017 for bipolar depression alone.
The Designation letter contains a specific finding that NRXP NRX-100 addresses an "unmet medical need." This is a specific qualifying requirement for the Commissioner's National Priority Voucher Program.
NRXP Filing of Commissioner's National Priority Voucher application for intravenous ketamine (NRX-100).
Submission of draft labeling for NRXP NRX-100 in the treatment of suicidal depression based on the Fast Track Designation received.
Filing of an Abbreviated New Drug Application (ANDA) for NRXP NRX-100 (preservative-free intravenous ketamine).
More on S For Story
Submission of stability data for NRXP NRX-100 to the manufacturing data on file with FDA sufficient to support three years of room temperature shelf stability for NRX-100.
Completion of a toxicology assessment of Benzethonium Chloride1, documenting its lack of "Generally Recognized as Safe" (GRAS) status and lack of safety data to support its use in intravenous presentations of ketamine.
NRXP filing of a Citizen's Petition with the U.S. Food and Drug Administration to seek the removal of benzethonium chloride, a toxic preservative, from all ketamine products for intravenous administration.
Filing of a patent application for NRXP NRX-100.
Receipt of a PDUFA filing fee waiver from the FDA for NRXP NRX-100.
NRXP filing of module 3 manufacturing data to support a New Drug Application for NRX-101 in the treatment of patients with suicidal bipolar depression and akathisia despite treatment with already-approved medication.
HOPE Therapeutics
NRXP execution of definitive Purchase Agreement and receipt of final regulatory clearance from Florida's Agency for Health Care Administration ("ACHA") to proceed with closing the acquisition of Dura Medical.
Execution of binding letter of intent to acquire the assets of NeuroSpa TMS Holdings of Tampa, FL.
Execution of a binding letter of intent to acquire a 49% interest in Cohen and Associates, LLC.
NRXP Receipt of approval, pending legal stipulations, for $7.8 million of debt financing to support the acquisition of Dura Medical, NeuroSpa TMS Holdings, and Cohen and Associates, LLC.
Corporate (subsequent to the filing of form 10-Q)
NRXP $6.5 million dollar investment to purchase approximately 3.9 million shares of common stock of NRx Pharmaceuticals on August 18, 2025, by a consortium of experienced biotechnology investors led by B Group Capital. The purchase is subject to a one-year lockup on trading, shorting, or otherwise hypothecating said securities. The investment has no warrants, repricing provisions, commissions, or other structure.
The B Group Capital led consortium of ultra long-term healthcare specialist investors is highly strategic with extensive experience in complex clinical, regulatory, and commercial therapeutics but also direct ownership and management of multi-unit retail operations with potentially positive long-term implications for efforts to continue to scale and develop NRXP HOPE Therapeutics.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
H.C. Wainright Analyst Report Cites Paradigm Shift in the Treatment of Depression With Suicidality; Assuming Coverage with Buy and $40 Price Target.
Dura Medical Acquisition Completed in Network of Interventional Psychiatry Clinics.
FDA Fast Track Designation for NRX 100 for Suicidal Ideation in Patients with Depression, Including Bipolar Depression.
Designation Includes an FDA Determination That NRX-100 has Potential to Address an Unmet Need.
Actions Taken to Request the Removal of Benzethonium Chloride from Ketamine Products in Favor of the Company's Safer and Superior Options.
$7.8 Million Debt Financing to Fuel NRXP HOPE Clinic Acquisitions with
Universal Capital, LLC.
Accepted Non-Binding Potential Terms to License and Distribute NRX-100 Drug Providing Over $300 Million in Milestones Plus Tiered Double-Digit Royalties.
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
On September 8th H.C. Wainwright has issued a new Analyst Report on NRXP: "A Paradigm Shift in the Treatment of Depression With Suicidality" Assuming Coverage With a Buy and $40 Price Target. The full report may be accessed at this direct link: https://hcwco.bluematrix.com/links2/secure/pdf/acdd3260-630e-48e6-9c2f-03fbc0be37d6
More on S For Story
- Amazon Bestselling Author, Apostle Tonya releases autographed copies of "Wonder & Glory"
- LSC Destruction Launches Cutting-Edge Cryptocurrency Scanning to Hard Drive Destruction Services
- No Better Time Than Now to Publish A Life Story
- History Matters: Book Recommendations for October
- $150 Million Financing Initiates N A S D A Q's First Tether Gold Treasury Combining the Stability of Physical Gold with Blockchain $AURE
Dura Medical Acquisition Completed in Network of Interventional Psychiatry Clinics
On September 8th NRXP announced the closing of its acquisition of Dura Medical. Dura, together with the pending Neurospa TMS and Cohen and Associates acquisitions, are planned to provide a comprehensive service offering to patients at more than 8 locations along the West Coast of Florida. Dura is revenue generating and EBITDA positive.
Dura delivers a full range of precision psychiatry services for severe depression and PTSD, including Ketamine Therapy and Transcranial Magnetic Stimulation to Veterans and civilian patients.
Expanded Access Policy for NRX-100 (preservative-free ketamine)
On August 27th NRXP announced its expanded access policy for NRX-100 (preservative-free ketamine) based on grant of Fast Track designation for NRX-100 in the treatment of suicidal ideation in patients with depression, including bipolar depression.
In granting the Fast Track designation, FDA made the determination that NRXP NRX-100 has the potential to address an unmet need, based on an assessment of the preliminary data contained in the Fast Track designation request. Accordingly, NRXP NRX-100 is available for expanded access to eligible patients.
Second Quarter 2025 Update
On August 18th NRXP announced financial results for the quarter ended June 30, 2025, and provided a corporate update. As of June 30, 2025, NRXP had approximately $2.9 million in cash and cash equivalents.
Drug Development
Grant of expanded Fast Track Designation for NRXP NRX-100 from the FDA for all indications and types of depression and related disorders based on its potential to satisfy an unmet medical need.
Approximately 10-fold expansion of the addressable market to 13 million Americans, compared to the original Fast Track Designation issued in 2017 for bipolar depression alone.
The Designation letter contains a specific finding that NRXP NRX-100 addresses an "unmet medical need." This is a specific qualifying requirement for the Commissioner's National Priority Voucher Program.
NRXP Filing of Commissioner's National Priority Voucher application for intravenous ketamine (NRX-100).
Submission of draft labeling for NRXP NRX-100 in the treatment of suicidal depression based on the Fast Track Designation received.
Filing of an Abbreviated New Drug Application (ANDA) for NRXP NRX-100 (preservative-free intravenous ketamine).
More on S For Story
- Podcast for Midlife Women Entrepreneurs Celebrates 100th Episode with Rhea Lana's Founder and CEO
- What If Help Could Come Before the Fall?
- Digi 995: Spookyverse — Unlocking the Multiverse of the Digiverse
- Fiction author re-imagines Acts of the Apostles with Female Apostles
- Combatting Commercial Clutter: Award-Winning Author Andrew Kooman Releases Gather for Advent
Submission of stability data for NRXP NRX-100 to the manufacturing data on file with FDA sufficient to support three years of room temperature shelf stability for NRX-100.
Completion of a toxicology assessment of Benzethonium Chloride1, documenting its lack of "Generally Recognized as Safe" (GRAS) status and lack of safety data to support its use in intravenous presentations of ketamine.
NRXP filing of a Citizen's Petition with the U.S. Food and Drug Administration to seek the removal of benzethonium chloride, a toxic preservative, from all ketamine products for intravenous administration.
Filing of a patent application for NRXP NRX-100.
Receipt of a PDUFA filing fee waiver from the FDA for NRXP NRX-100.
NRXP filing of module 3 manufacturing data to support a New Drug Application for NRX-101 in the treatment of patients with suicidal bipolar depression and akathisia despite treatment with already-approved medication.
HOPE Therapeutics
NRXP execution of definitive Purchase Agreement and receipt of final regulatory clearance from Florida's Agency for Health Care Administration ("ACHA") to proceed with closing the acquisition of Dura Medical.
Execution of binding letter of intent to acquire the assets of NeuroSpa TMS Holdings of Tampa, FL.
Execution of a binding letter of intent to acquire a 49% interest in Cohen and Associates, LLC.
NRXP Receipt of approval, pending legal stipulations, for $7.8 million of debt financing to support the acquisition of Dura Medical, NeuroSpa TMS Holdings, and Cohen and Associates, LLC.
Corporate (subsequent to the filing of form 10-Q)
NRXP $6.5 million dollar investment to purchase approximately 3.9 million shares of common stock of NRx Pharmaceuticals on August 18, 2025, by a consortium of experienced biotechnology investors led by B Group Capital. The purchase is subject to a one-year lockup on trading, shorting, or otherwise hypothecating said securities. The investment has no warrants, repricing provisions, commissions, or other structure.
The B Group Capital led consortium of ultra long-term healthcare specialist investors is highly strategic with extensive experience in complex clinical, regulatory, and commercial therapeutics but also direct ownership and management of multi-unit retail operations with potentially positive long-term implications for efforts to continue to scale and develop NRXP HOPE Therapeutics.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
Filed Under: Media
0 Comments
Latest on S For Story
- White Glove Restoration Sends "Help in Boxes" to Support Communities in Ghana
- Ontario Publisher Wins Silver in the 2025 Moonbeam Children's Book Awards for Best Picture Book
- NASA Agreements, New Ocean Exploration Applications Added to Partnerships with Defiant Space Corp and Emtel Energy USA for Solar Tech Leader: $ASTI
- MackinPublic Provides Content and Confidence To US Public Libraries
- Chic and Secure: The Blue Luna Debuts Stylish Keychains with Purpose
- BEC Technologies Showcases Leadership in Private Broadband Ecosystem with Inclusion in UTC's Ecosystem Summary Report
- $500,000 in Stock Dividend for Shareholders in 2025 Sweetens The Pot on Success of Becoming Debt Free with No Convertible Notes or Warrants for $IQST
- Milwaukee Job Corps Center: Essential Workforce Training—Admissions Now Open
- Aissist.io Launches Hybrid AI Workforce to Solve AI Pilot Failure for Customer Support Automation
- New Chapter Book Teaches Courage And The Importance Of Friendship Through Fantasy-Driven Adventure
- Christy Sports Makes Snowsports More Accessible for Families to Get Outside Together
- MainConcept Completes Management Buyout to Become Independent Company
- LIB Industry Expands Full-Series Salt Spray Corrosion Test Chambers to Meet Global Testing Standards
- The Easy Way to Collect Every Wedding Photo from Your Guests - No App Needed
- REPRESENTATION REVOLUTION: FLM TV Network Launches as America's First Truly Diverse Broadcast Network
- New Book Call all Jews to Accept Jesus as the Messiah
- For Nat'l. Novel Writing Month: Check out The Next Big Thing in Entertainment for Books, Movies & TV
- MetroWest wellness: Holliston farmhouse spa unveils Centerpoint Studio
- Cancer Survivor Roslyn Franken Marks 30-Year Milestone with Empowering Gift for Women Survivors
- "Grace & Grit" Helps Mompreneurs Live Boldly in Faith and Purpose